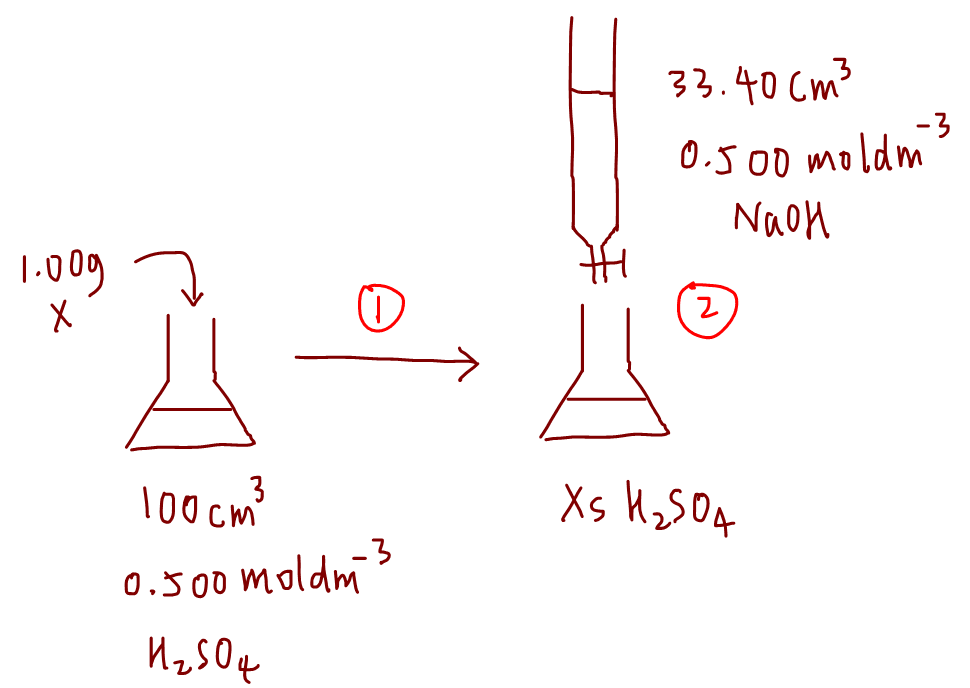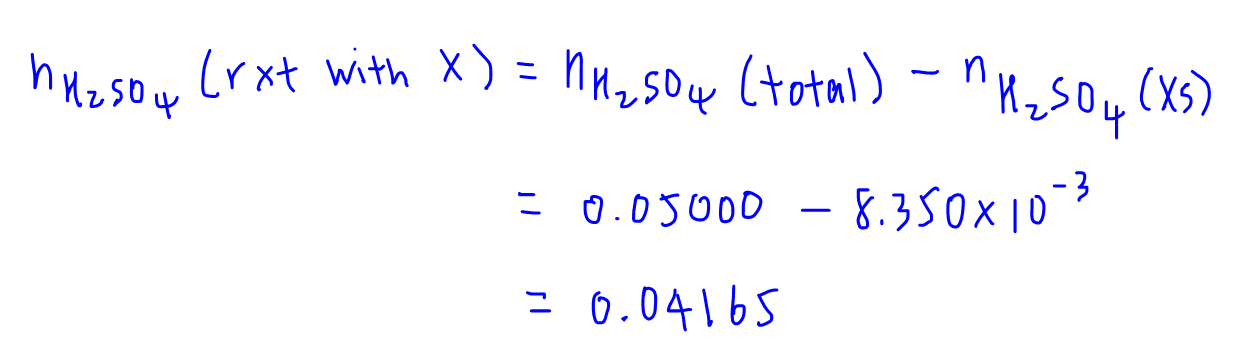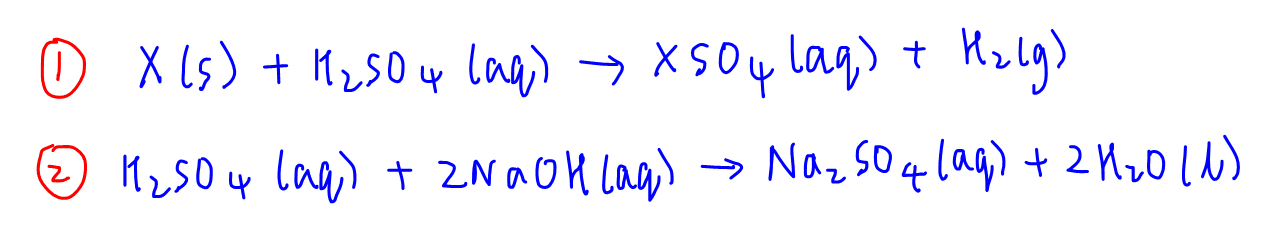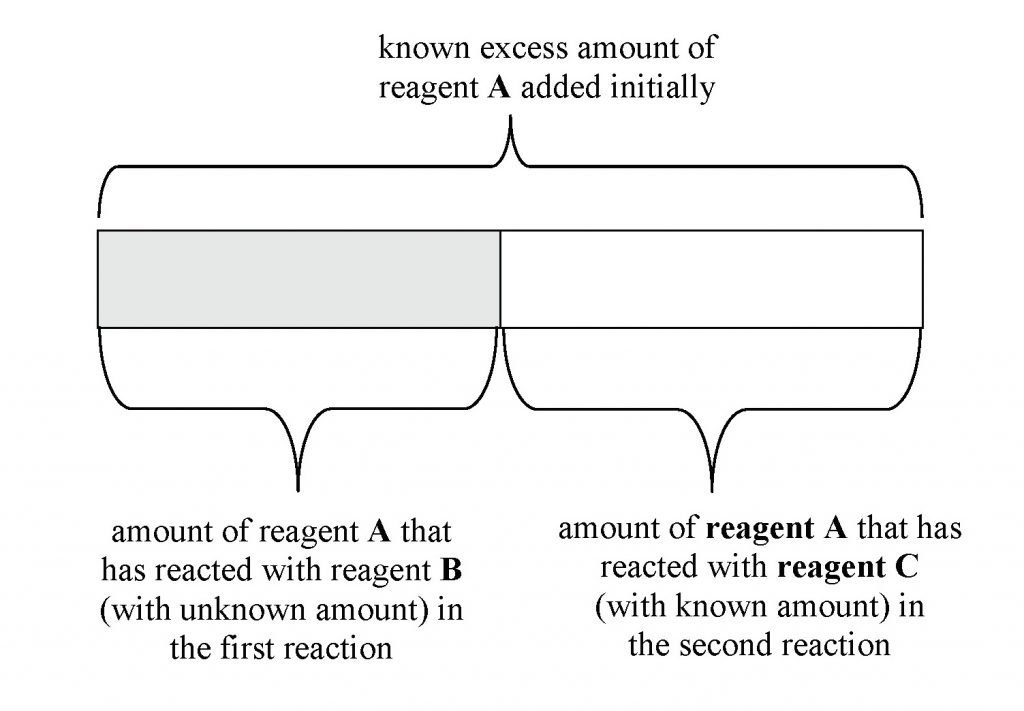
5 Simple Steps to Solve Back Titration Questions in Volumetric Analysis - A-Level H2 Chemistry Tuition by 10 Year Series Author

SOLVED: 9.In the back titration of aspirin, an excess amount of NaOH was reacted with aspirin and the excess was then back titrated with HCI. Suppose 0.4567 sample of aspirin was reacted