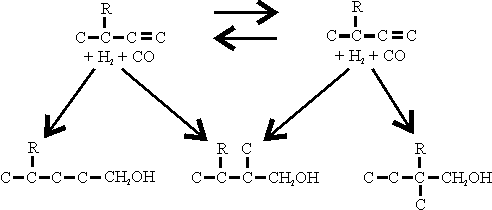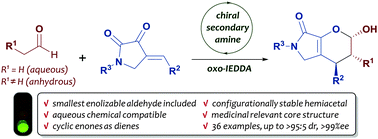
Asymmetric synthesis of bicyclic dihydropyrans via organocatalytic inverse-electron-demand oxo-Diels–Alder reactions of enolizable aliphatic aldehydes - Chemical Communications (RSC Publishing)

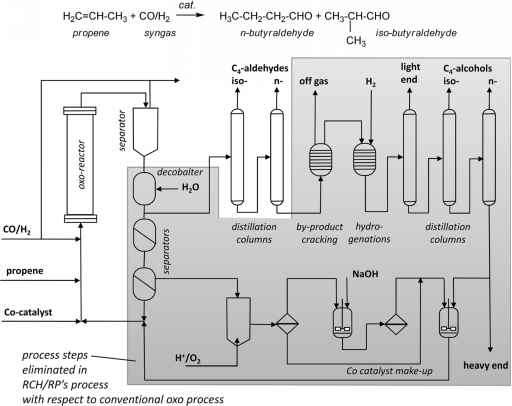
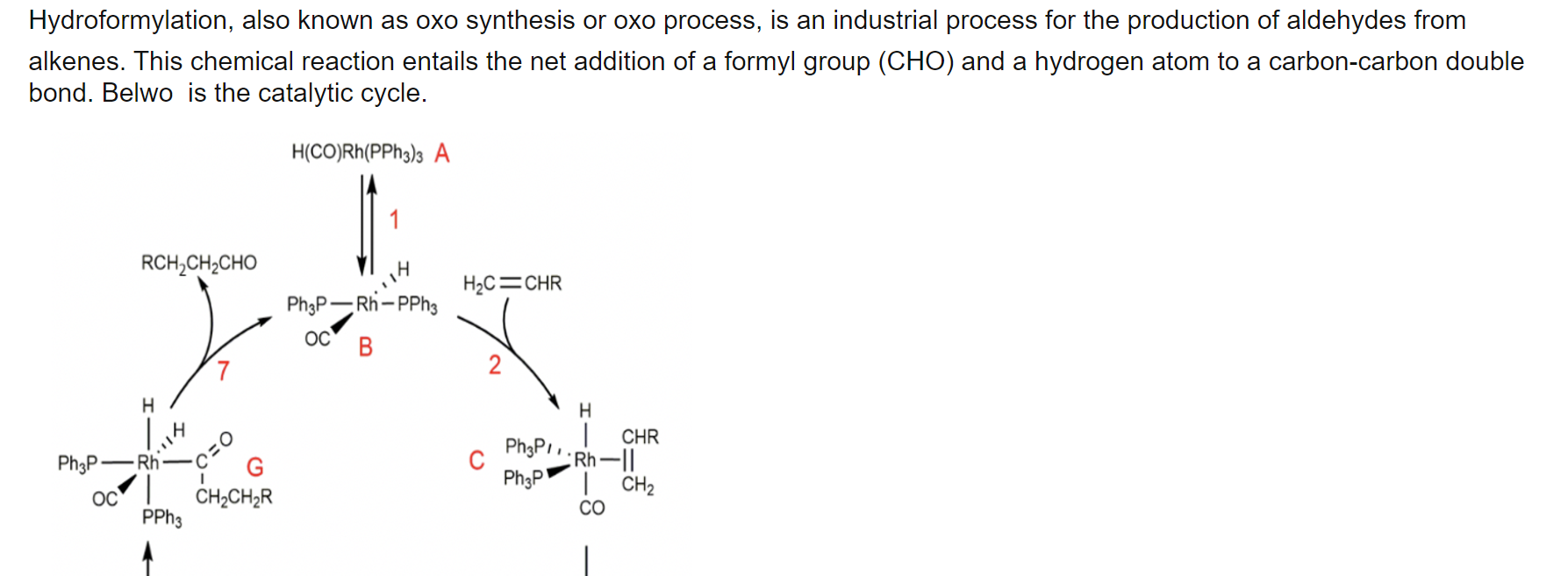
Figure 2. Scheme of the Ruhrchemie/ Rhône-Poulenc oxo process. a) reaction scheme and structure of the rhodium catalyst. b) Formation of biphasic liquid mixture that is split in a separator to obtain
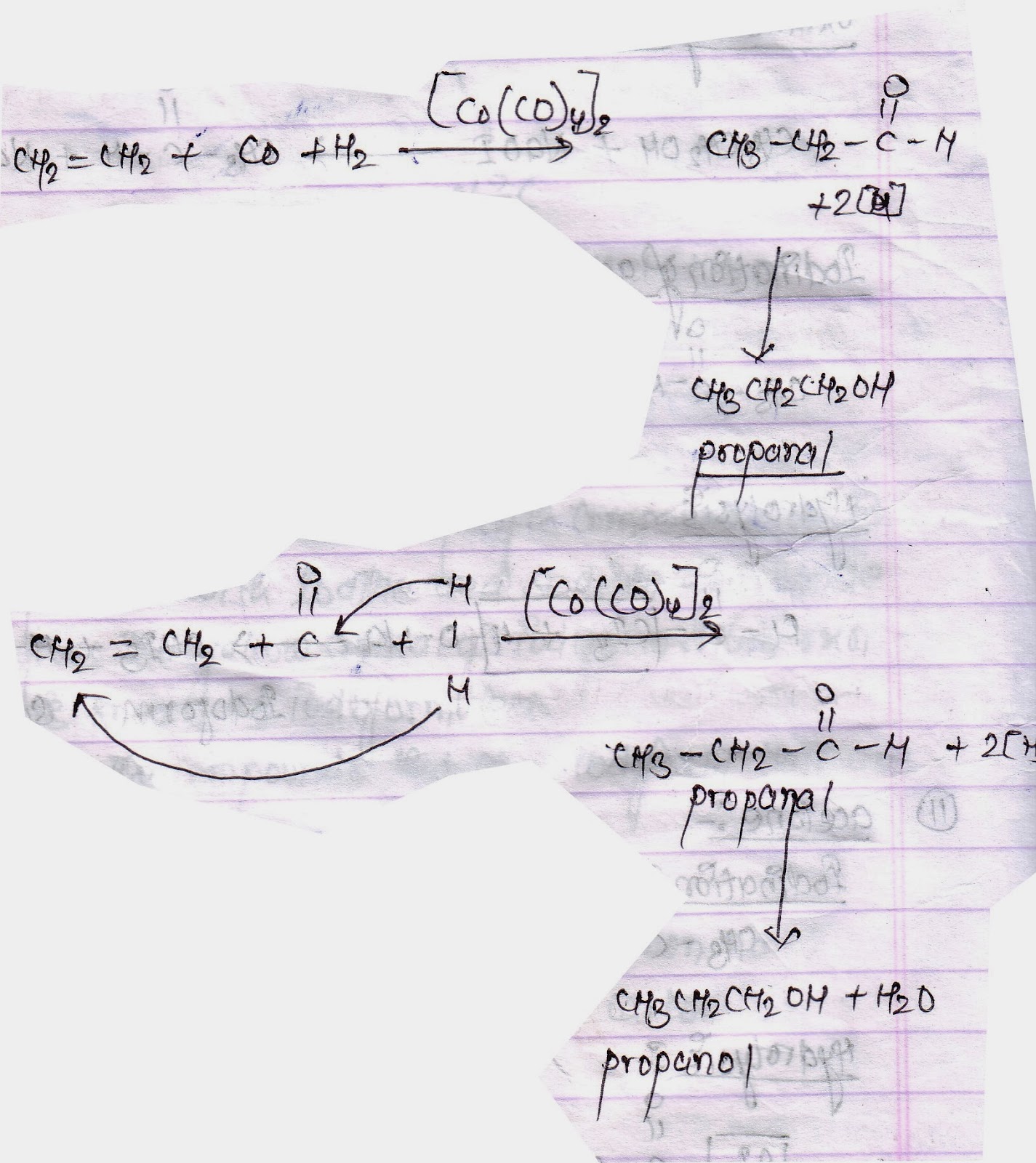
Explain in detail the mechanism (along with the role of catalyst) of Wacker process and oxo process for the synthesis of aldehydes and ketones





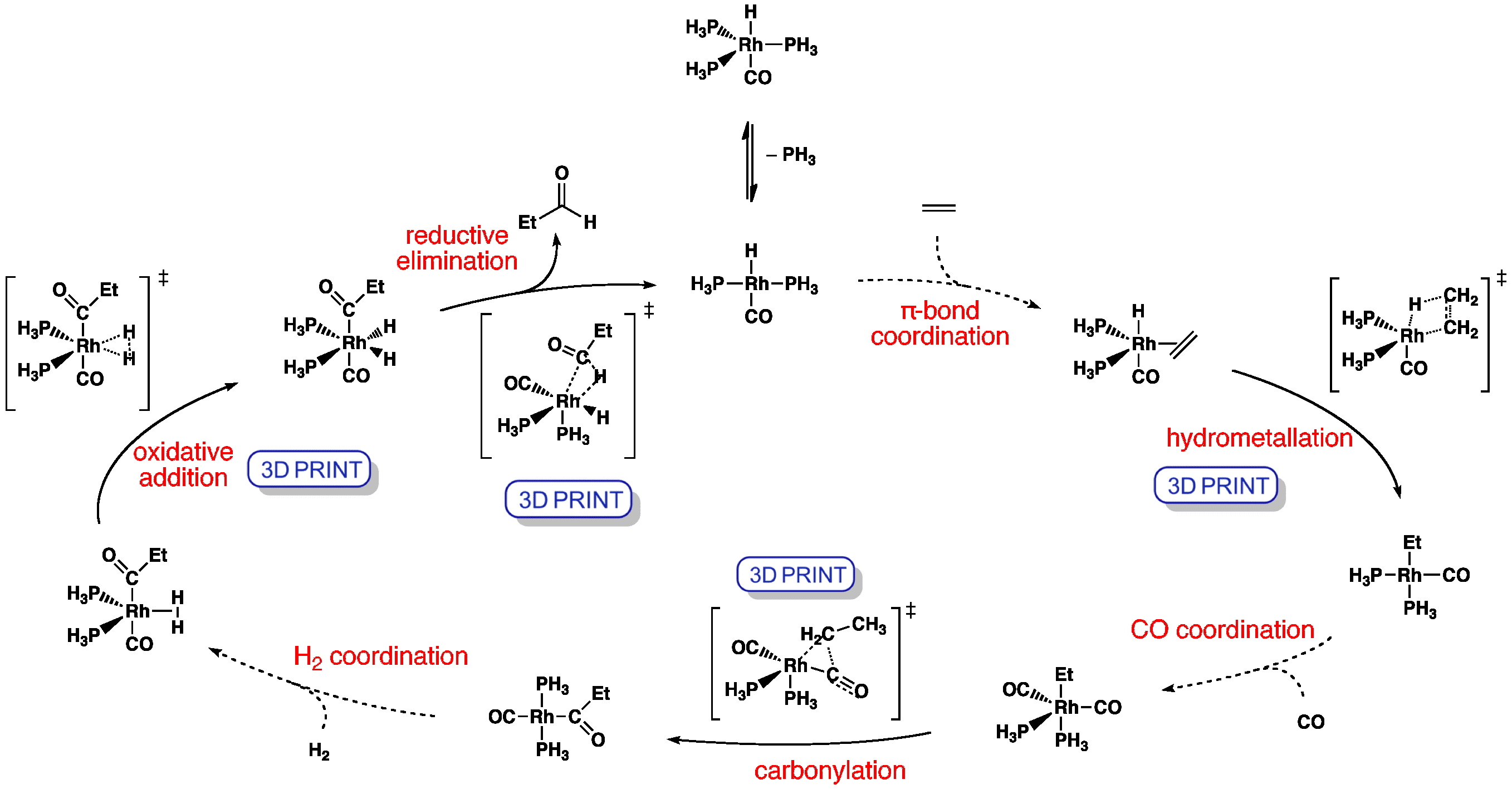
![Hydroformylation reaction [oxo process ] - YouTube Hydroformylation reaction [oxo process ] - YouTube](https://i.ytimg.com/vi/vnVMS_Dp0dU/hqdefault.jpg)