![The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system | SpringerLink The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs11051-011-0495-8/MediaObjects/11051_2011_495_Fig6_HTML.gif)
The effect of [Fe3+]/[Fe2+] molar ratio and iron salts concentration on the properties of superparamagnetic iron oxide nanoparticles in the water/ethanol/toluene system | SpringerLink

Figure 3 from First-Principles Fe L2,3-Edge and O K-Edge XANES and XMCD Spectra for Iron Oxides. | Semantic Scholar

Iron transition metal Chemistry iron(II) Fe2+ iron(III) Fe3+ complexes ions ligand substitution redox chemical reactions principal oxidation states +2 +3 extraction GCE AS A2 IB A level inorganic chemistry revision notes

Iron transition metal Chemistry iron(II) Fe2+ iron(III) Fe3+ complexes ions ligand substitution redox chemical reactions principal oxidation states +2 +3 extraction GCE AS A2 IB A level inorganic chemistry revision notes

Figure 1 from Electronic spectra of Fe 3 + oxides and oxide hydroxides in the near IR to near UV | Semantic Scholar

Manetite, which has the formula Fe(2)O(3) is comprised or iron(II) oxide and iron (III) oxide. What is the ratio of iron(II) ions to iron(III) ions in magnetiude.

Iron transition metal Chemistry iron(II) Fe2+ iron(III) Fe3+ complexes ions ligand substitution redox chemical reactions principal oxidation states +2 +3 extraction GCE AS A2 IB A level inorganic chemistry revision notes
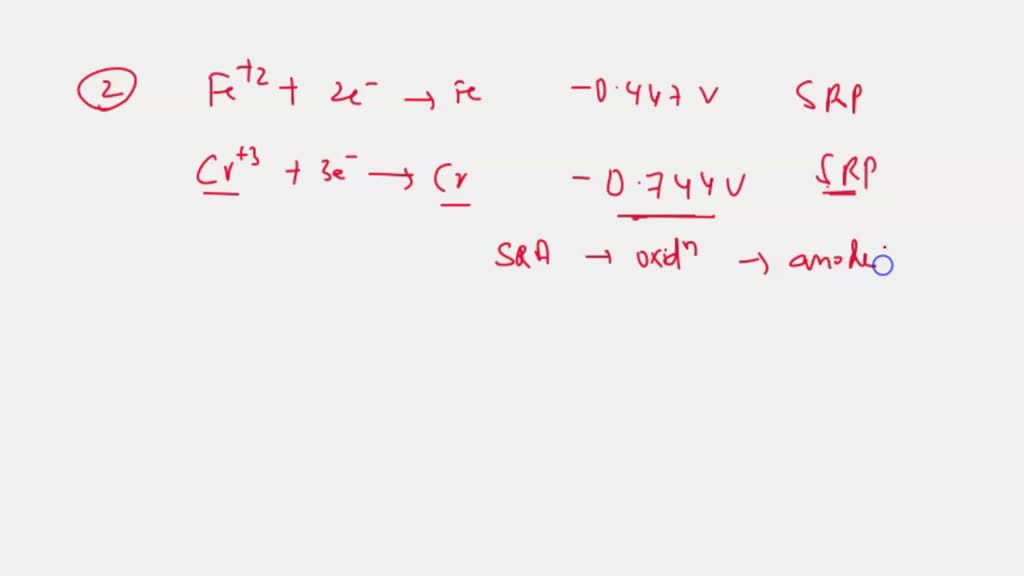
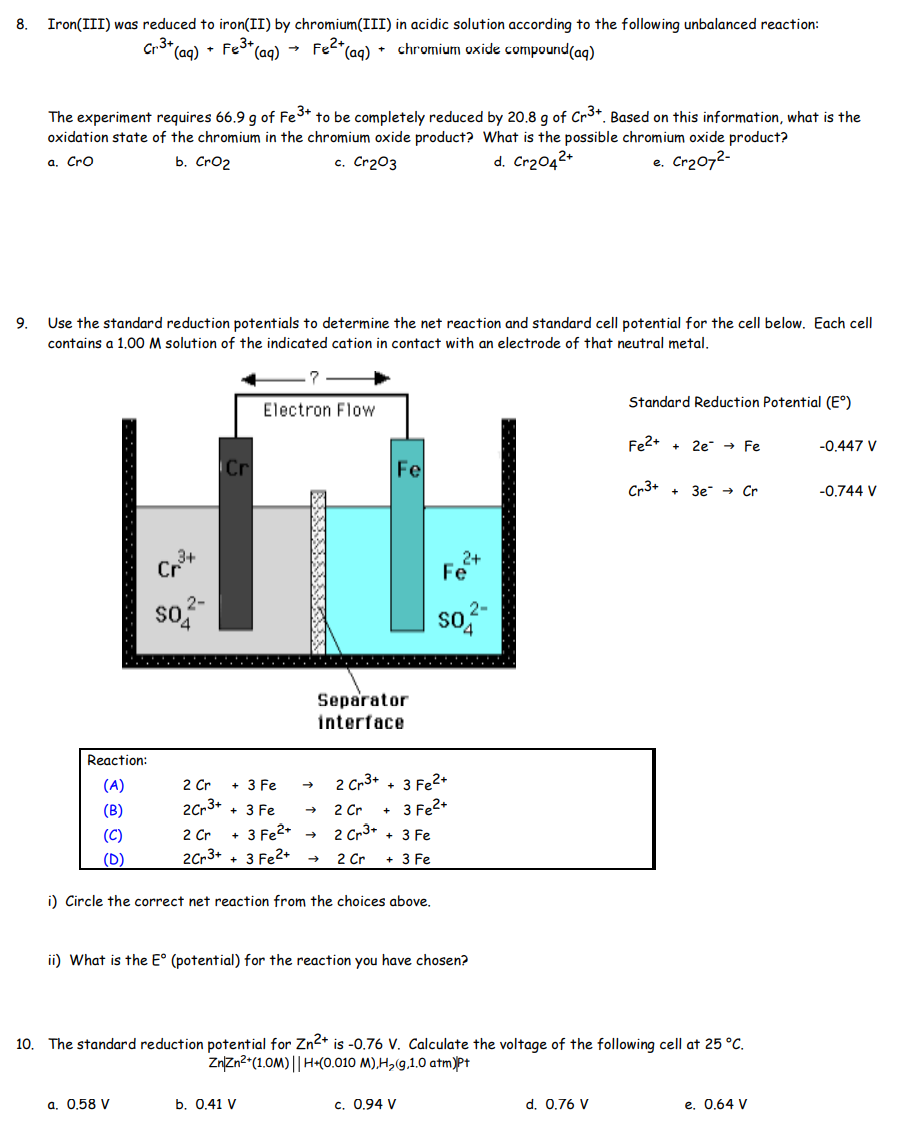
SOLVED: Iron(III) was reduced to iron(II) by chromium(III) in acidic solution according to the following unbalanced reaction: Cr3+(aq) + Fe3+(aq) ⟷ Fe2+(aq) + chromium oxide compound(aq) The experiment requires 66.9 g of
If Fe2+ and Fe3+ are present in Oxide of Iron Fe0.78O0.96 .Then Calculate Number of These Isotopes in Iron Oxide and Their

Ocean productivity before about 1.9 Gyr ago limited by phosphorus adsorption onto iron oxides | Nature

The Estimation of Iron (II) and Iron (III) in a Mixture Containing Both - Free comparison essay example, compare and contrast paper

Iron oxide (Fe0.94O) exist. What reaction of iron ions i.e. Fe^2 + and Fe^3 + present in this? Calculate their percentage.

Electrochemical Analysis of Changes in Iron Oxide Reducibility during Abiotic Ferrihydrite Transformation into Goethite and Magnetite | Environmental Science & Technology

Gold, Silver, and Iron Oxide Nanoparticle Incorporation into Silk Hydrogels for Biomedical Applications: Elaboration, Structure, and Properties | ACS Biomaterials Science & Engineering

Photo-catalytic selectivity of anthranilic acid over iron oxide incorporated titania nanoparticles: Influence of the Fe2+/Fe3+ ratio of iron oxide - ScienceDirect


![A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0304885317309411-gr4.jpg)

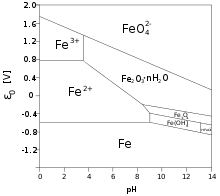
![A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0304885317309411-gr8.jpg)
![A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect A method for determination of [Fe3+]/[Fe2+] ratio in superparamagnetic iron oxide - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0304885317309411-gr1.gif)