
POL-0030: Compliance and enforcement approach and inspection strategy for clinical trials of drugs involving human subjects - Canada.ca
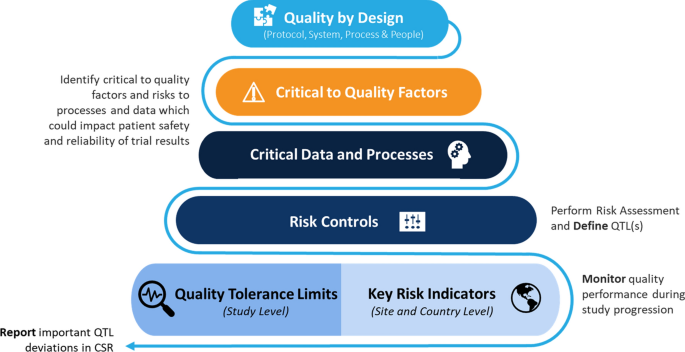
Quality Tolerance Limits: Framework for Successful Implementation in Clinical Development | Therapeutic Innovation & Regulatory Science













