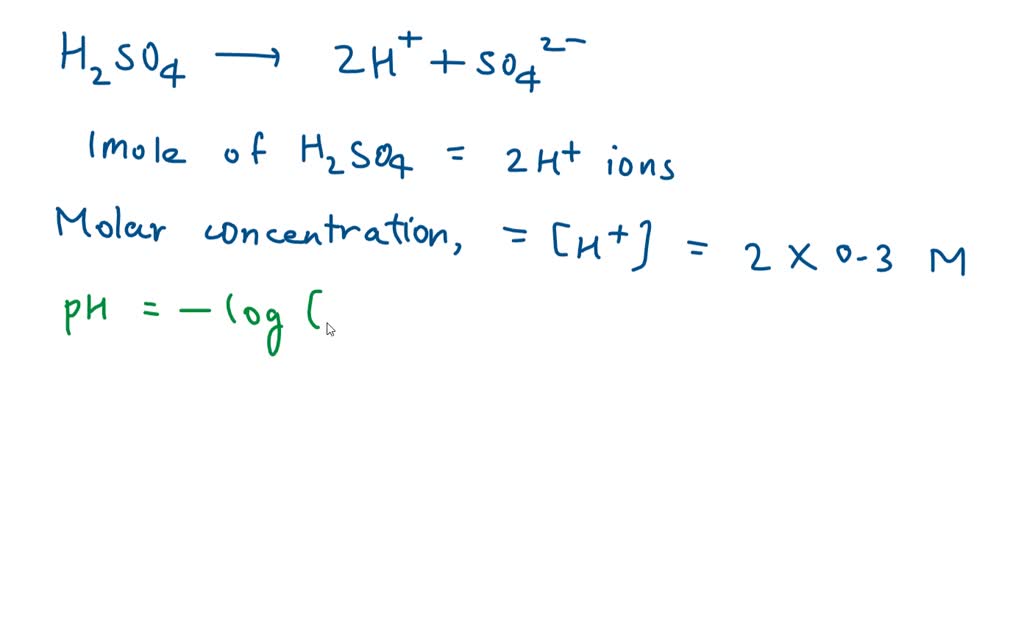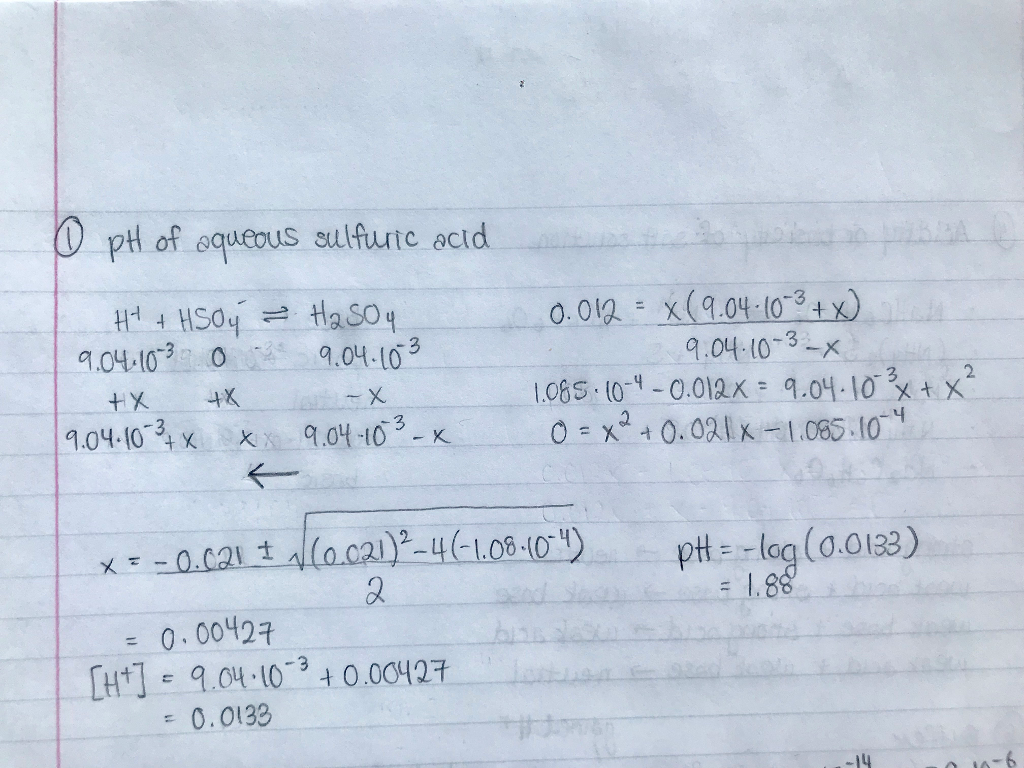Dissociation Constants of Perchloric and Sulfuric Acids in Aqueous Solution | The Journal of Physical Chemistry B

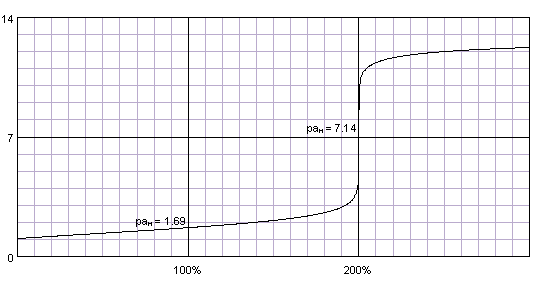
Curve for pH as a function of sulfuric acid concentration based on the... | Download Scientific Diagram
![SOLVED: Find the pH of a 0.0065 M sulfuric acid solution. Find [SO42?] of a 0.0065 M sulfuric acid solution. Answer in M SOLVED: Find the pH of a 0.0065 M sulfuric acid solution. Find [SO42?] of a 0.0065 M sulfuric acid solution. Answer in M](https://cdn.numerade.com/ask_previews/5be48cb9-2b9b-4861-a0b2-0ce3c9e0c22f_large.jpg)
SOLVED: Find the pH of a 0.0065 M sulfuric acid solution. Find [SO42?] of a 0.0065 M sulfuric acid solution. Answer in M

Spectroscopic Measurement of pH in Aqueous Sulfuric Acid and Ammonia from Sub- to Supercritical Conditions | Industrial & Engineering Chemistry Research
![SOLVED: Calculate the pH of a 0.1wt% H2SO4 SULFURIC ACID solution. You can assume that the density of the solution is that of water. Must use ph = -log[h+]. Please explain and SOLVED: Calculate the pH of a 0.1wt% H2SO4 SULFURIC ACID solution. You can assume that the density of the solution is that of water. Must use ph = -log[h+]. Please explain and](https://cdn.numerade.com/ask_previews/fb910f96-3dc9-421e-9ac5-8d5a0aaf4d70_large.jpg)