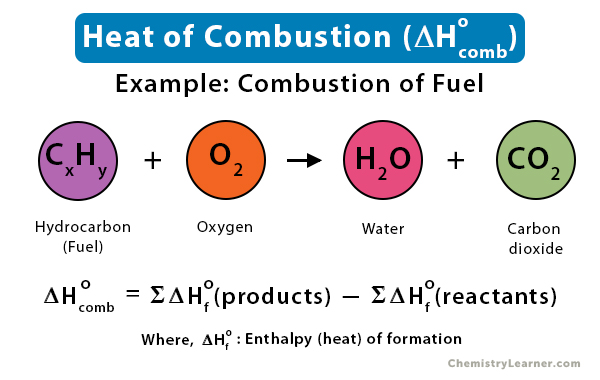
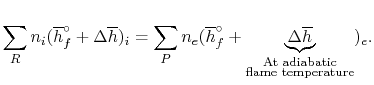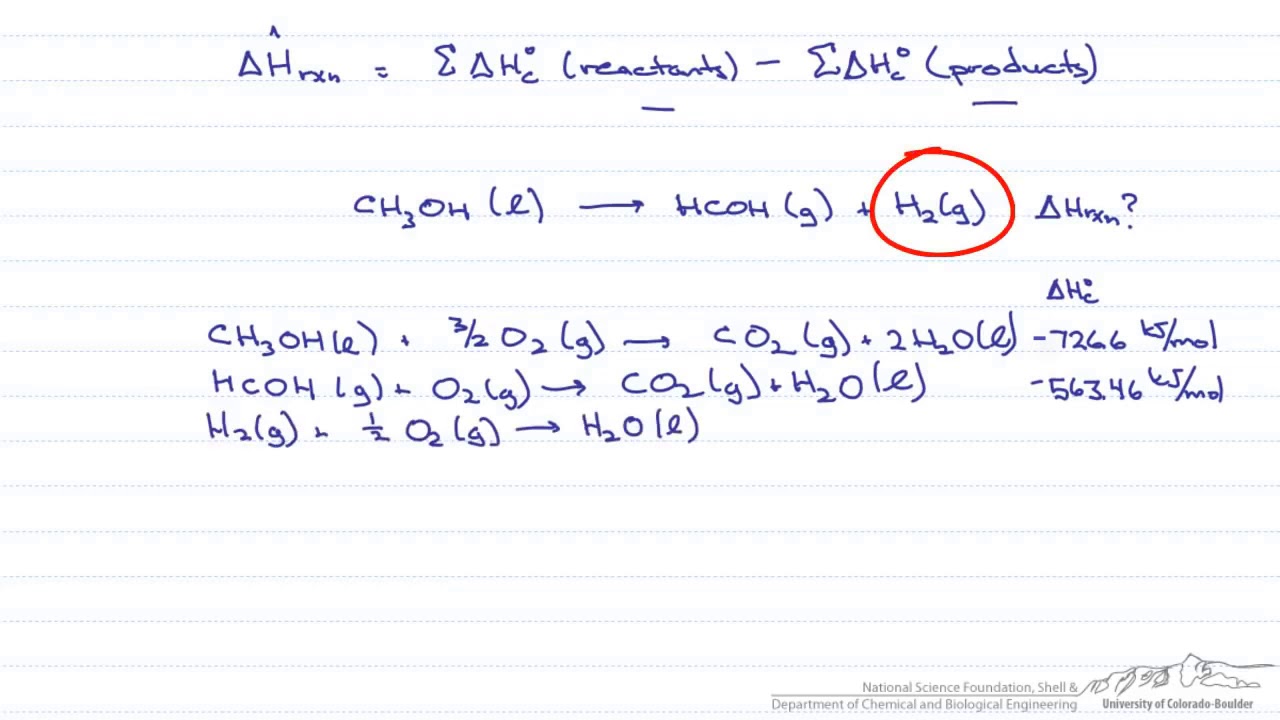![Total: 2 Average: 5/5] What is the heat of combustion? What is the definition of enthalpy of c… | Chemistry lessons, Chemistry classroom, Chemistry notes Total: 2 Average: 5/5] What is the heat of combustion? What is the definition of enthalpy of c… | Chemistry lessons, Chemistry classroom, Chemistry notes](https://i.pinimg.com/736x/99/83/b8/9983b8be657f09567f1e4b88e6bed700.jpg)
Total: 2 Average: 5/5] What is the heat of combustion? What is the definition of enthalpy of c… | Chemistry lessons, Chemistry classroom, Chemistry notes

Combustion Calculations Calculate the theoretical flame temperature of combustion of hydrogen with theoretical amount of air assuming (a)No dissociation. - ppt download

Question Video: Calculating Standard Enthalpy of Combustion of Methane Using Standard Enthalpies of Formation of Methane and Carbon Dioxide | Nagwa

From the following data of heats of combustion, find the heat of formation of CH3OH(l) : CH3OH(l) + 32O2(g)⟶ CO2(g) + 2H2O(l);Δ H = - 726kJ C(s) + O2(g)⟶ CO2(g);Δ H = -

Calculate heat of combustion of coal, when 0.5kg of coal on burning raise the temperature of 50 - YouTube

propulsion - How do you determine what the temperature will be in the combustion chamber of a rocket engine? - Space Exploration Stack Exchange