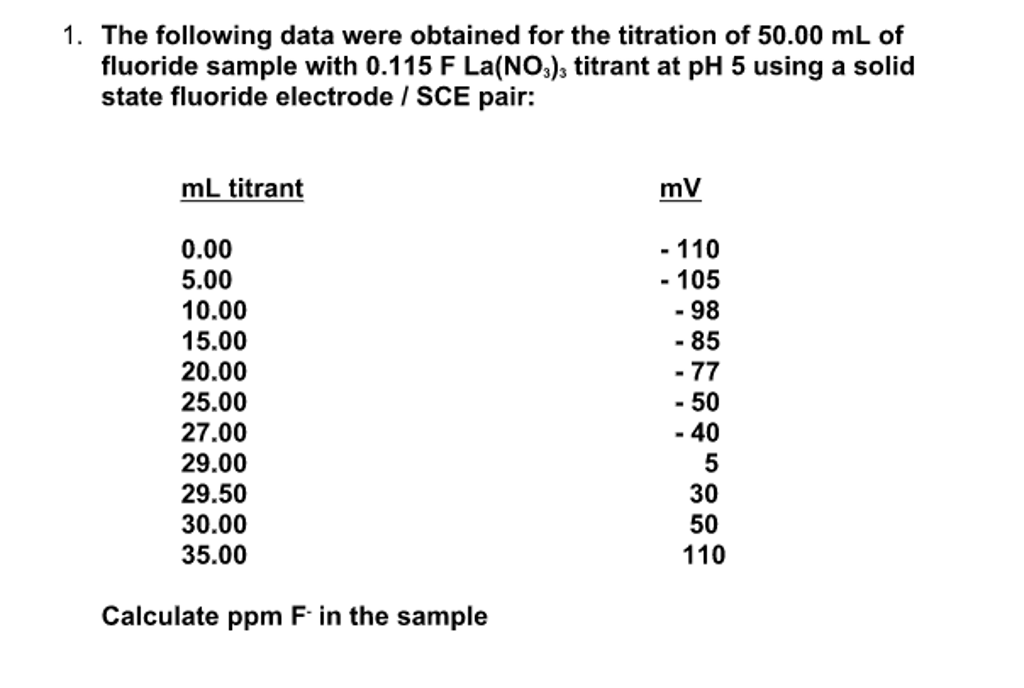
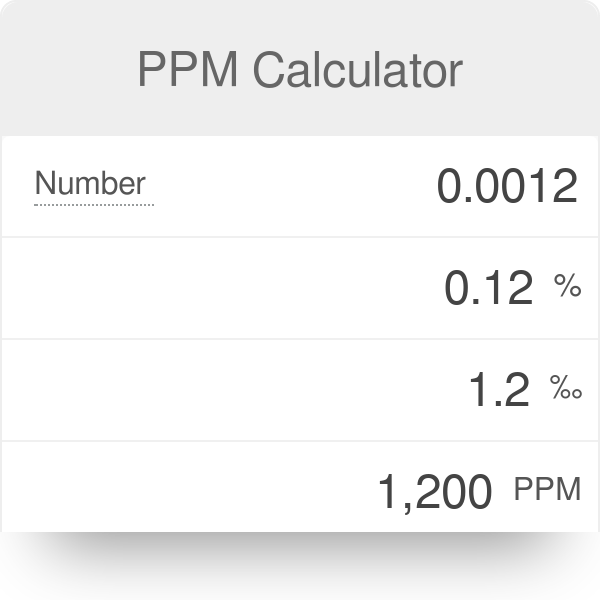
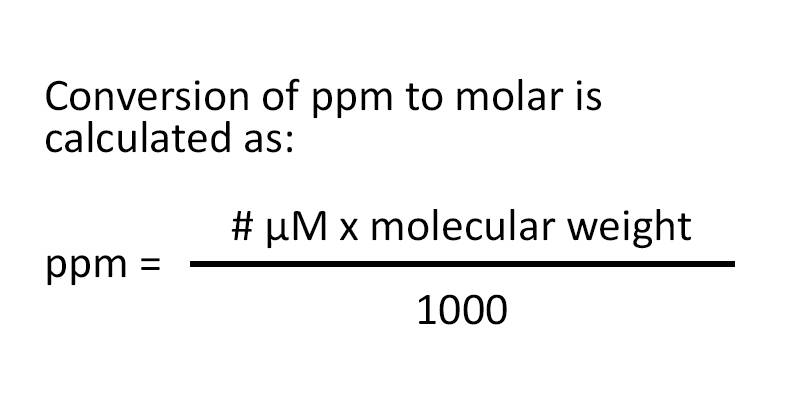
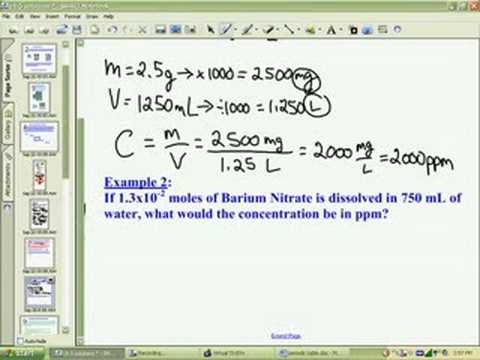
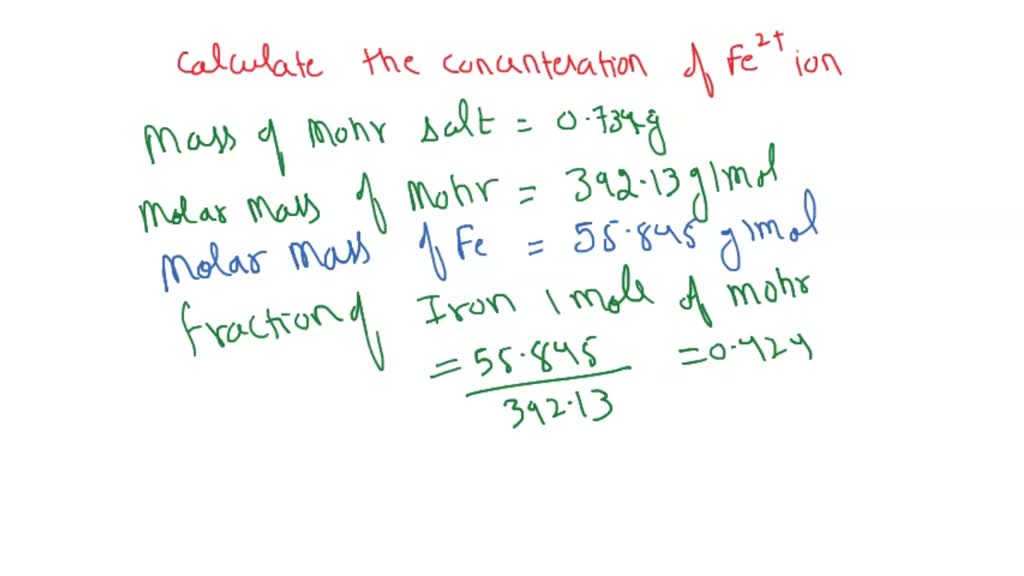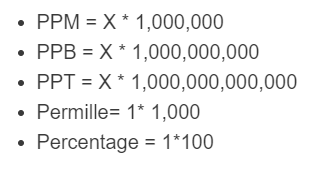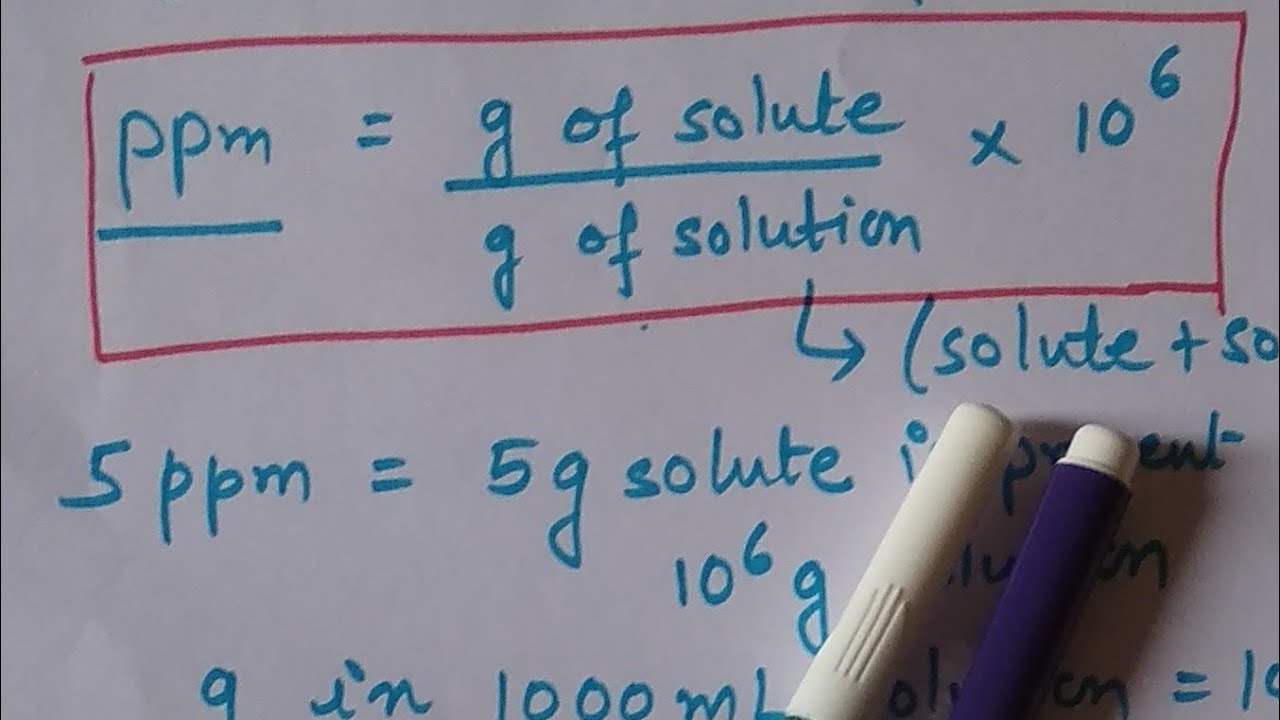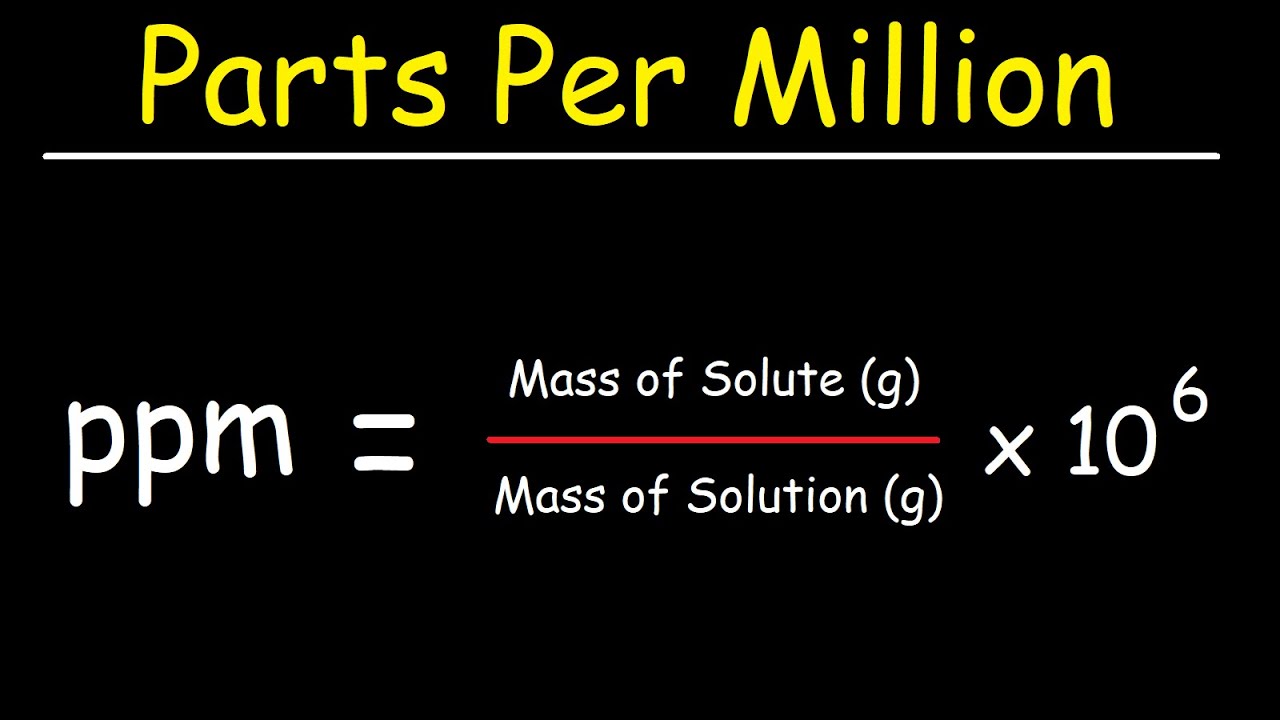SOLVED: In clinical applications, the unit parts per million (ppm) is used to express very small concentrations of solute, where ppm equivalent to mg of solute per 1 L of solution. Calculate

What is the formula to calculate the actual results of a diluted and digested sample by using AAS in PPM | ResearchGate