What is the molarity of a final solution after mixing 30 ml of 0.2M HCL and 20 ml of a 0.1M naoh solution? - Quora

What will be the molarity of the solution in which 0.365 g of HCl gas is dissolved in 100 mL of solution?

Calculate the molarity of a solution that is 39.77% H2SO4 by mass. The specific gravity of the solution is 1.305. | Homework.Study.com

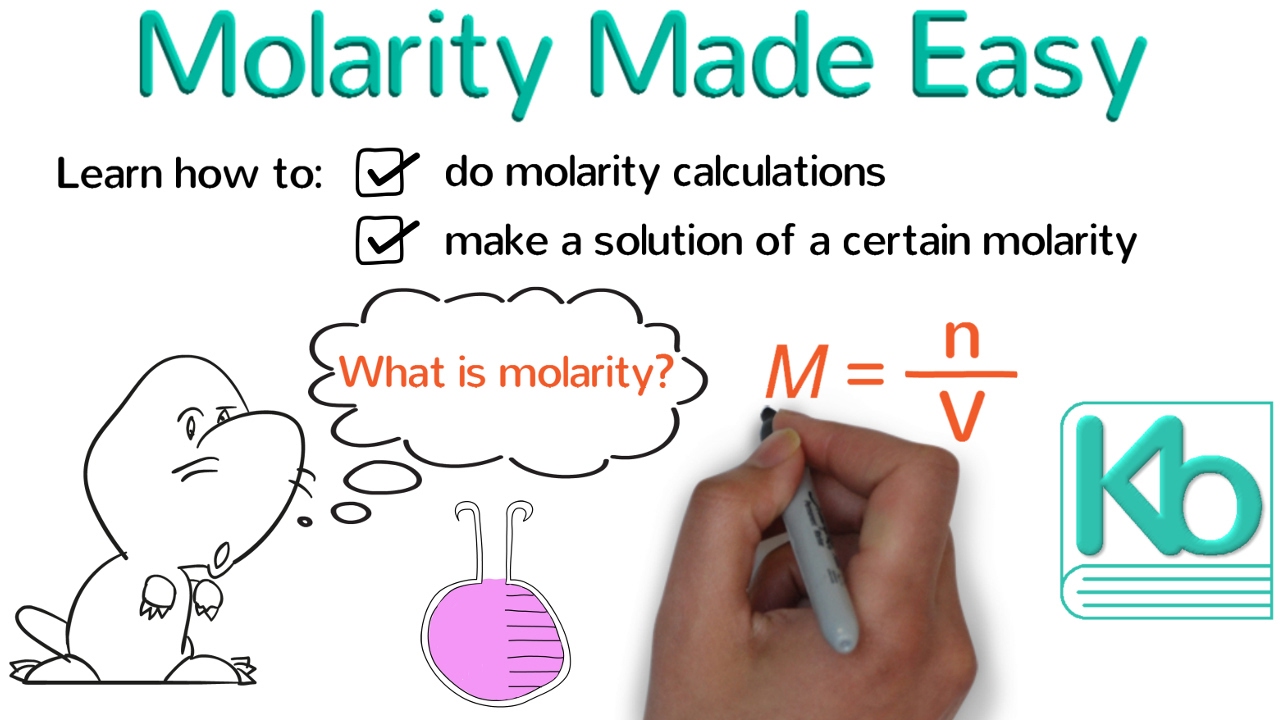
Molarity, Molality, Volume & Mass Percent, Mole Fraction & Density - Solution Concentration Problems - YouTube









:max_bytes(150000):strip_icc()/606823-calculate-molarity-of-a-solution-FINAL-5b7d7e15c9e77c0050355d4e.png)






