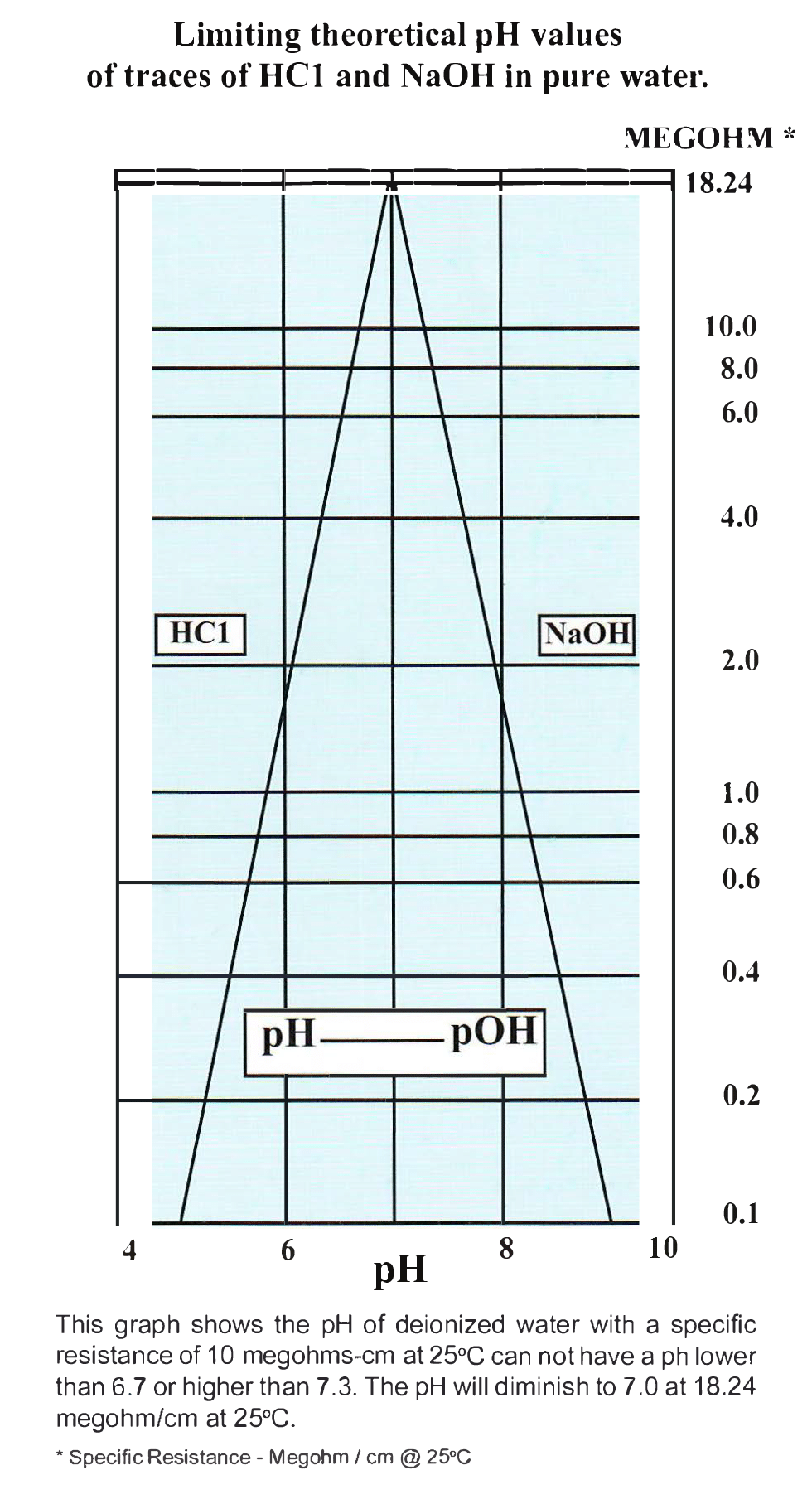
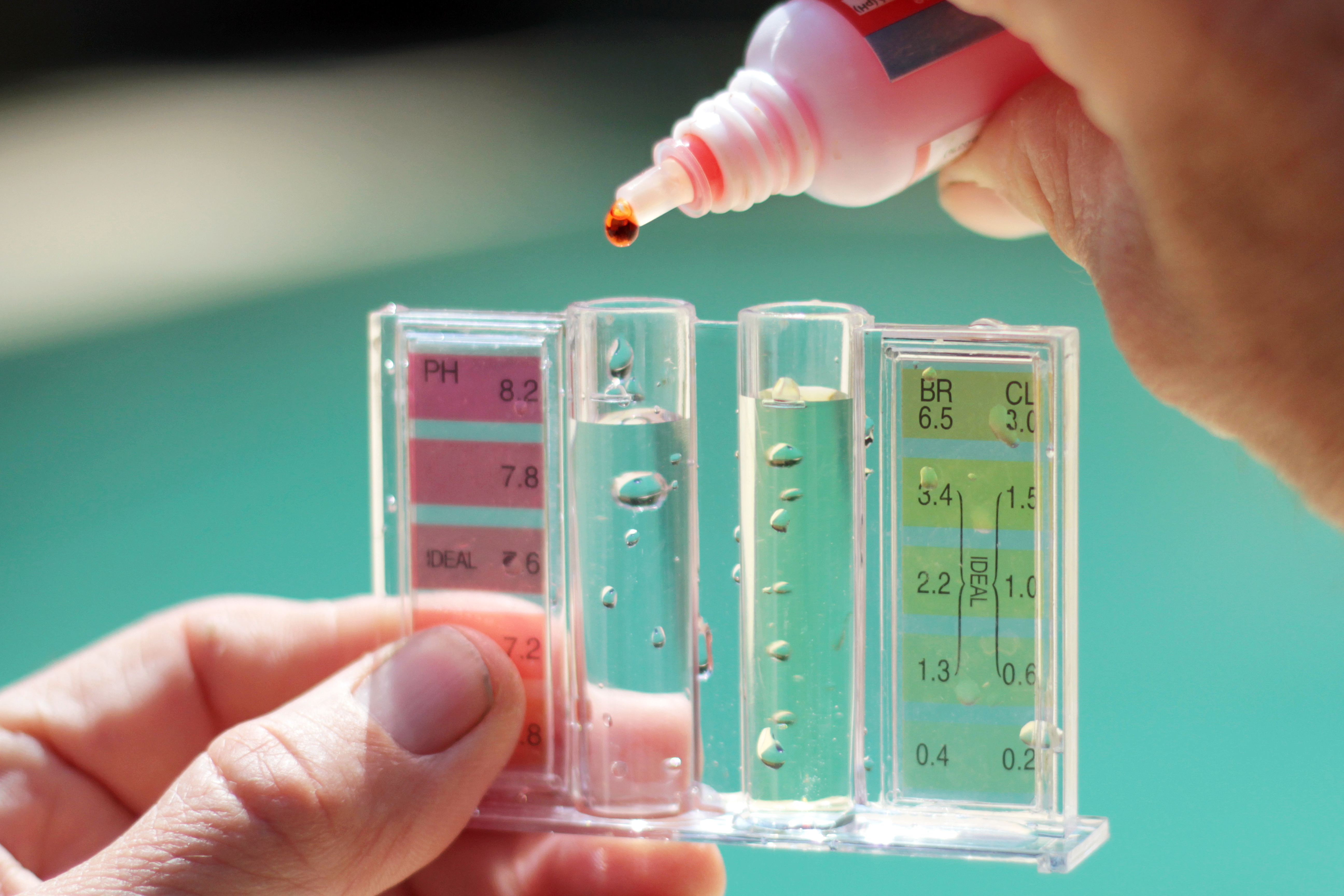
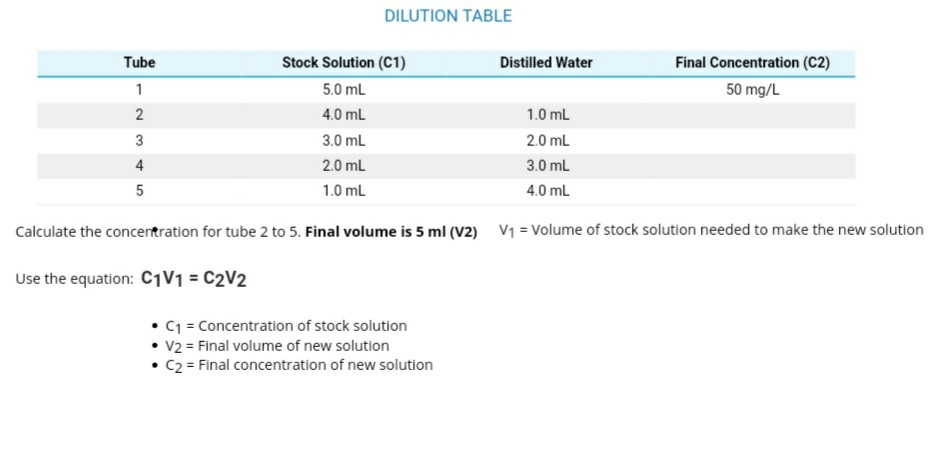
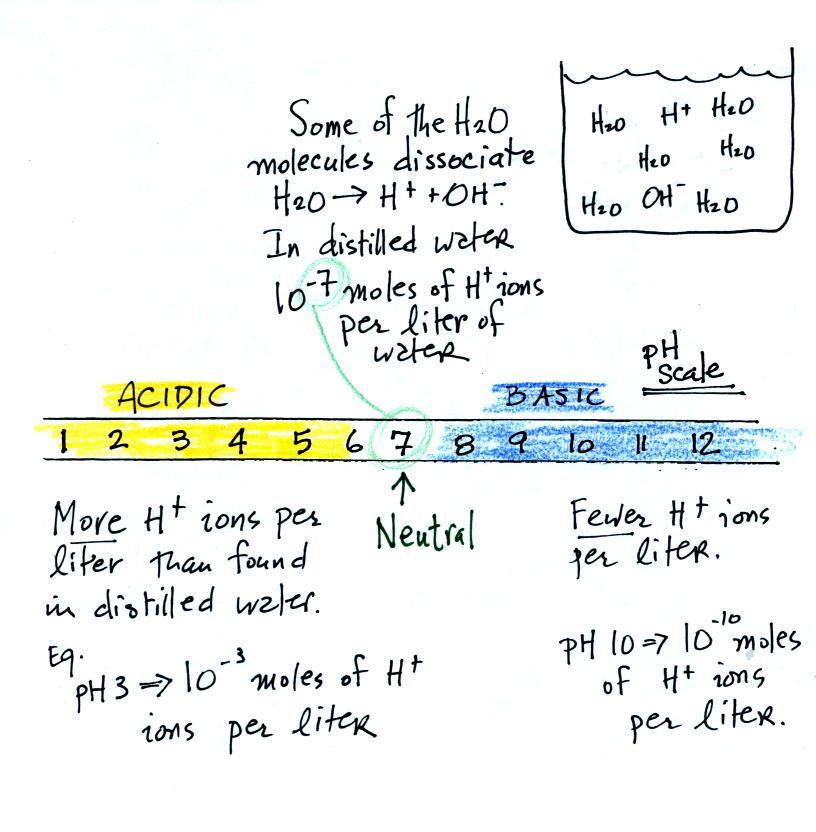
pHgraduatedscale.jpg - Concentration of Hydrogen ions compared to distilled water Examples 10 000 000 PH 0 Battery acid 1 000 000 PH 1 Hydrochloric | Course Hero

Concentration of magnesium (mg L−1) in distilled water and artificial... | Download Scientific Diagram

The concentration of metals (ppm) in double distilled water at 60±2ºC... | Download Scientific Diagram
![At 80^(@)C distilled water has [H(3)O^(+)] concentration equal [OH^(-)] 1xx10^(-6) "mole"//litre. The value of K(w) at this temperature will be At 80^(@)C distilled water has [H(3)O^(+)] concentration equal [OH^(-)] 1xx10^(-6) "mole"//litre. The value of K(w) at this temperature will be](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/52405125_web.png)
At 80^(@)C distilled water has [H(3)O^(+)] concentration equal [OH^(-)] 1xx10^(-6) "mole"//litre. The value of K(w) at this temperature will be