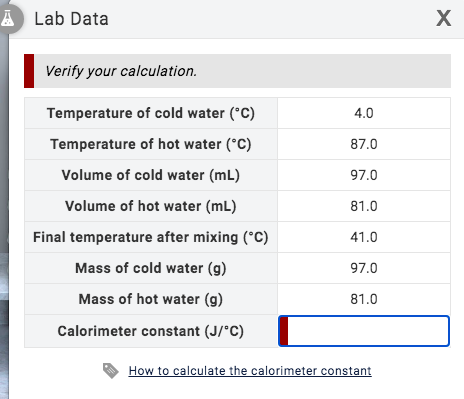
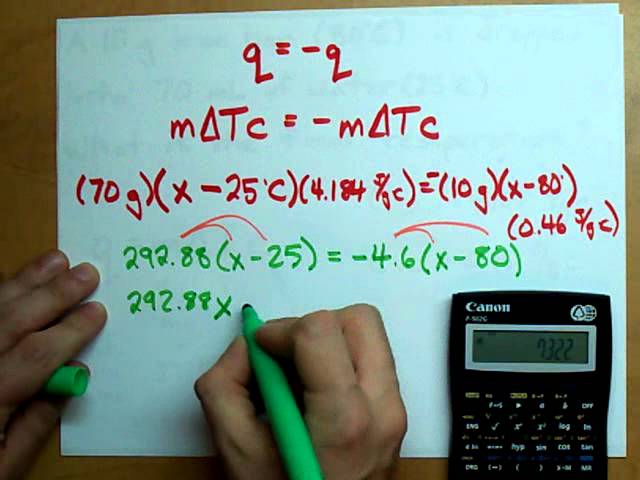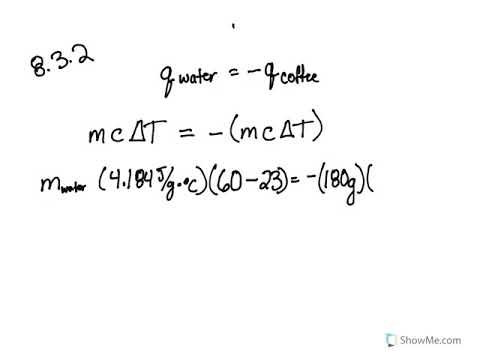SOLVED: Results: Calculate the heat gained by the water (lost by the substance in the calorimeter using the equation in the Introduction: Remember t0 write the unitsl! Q fon water using Ist

Question Video: Calculating the Heat Energy Transferred to Water Using Its Specific Heat Capacity | Nagwa

ThermoChem Class #4 – Calorimetry OB: how is energy in food measured? (It's the “bomb”!) You will need a calculator and your thinking hat now. - ppt download

ThermoChem Class #4 – Calorimetry OB: how is energy in food measured? (It's the “bomb”!) You will need a calculator and your thinking hat now. - ppt download