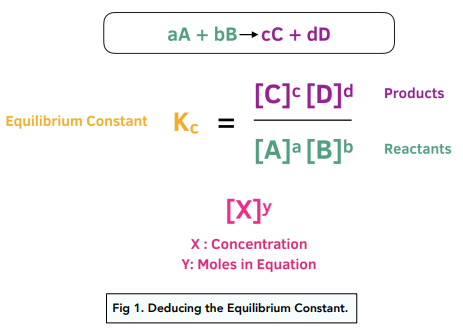
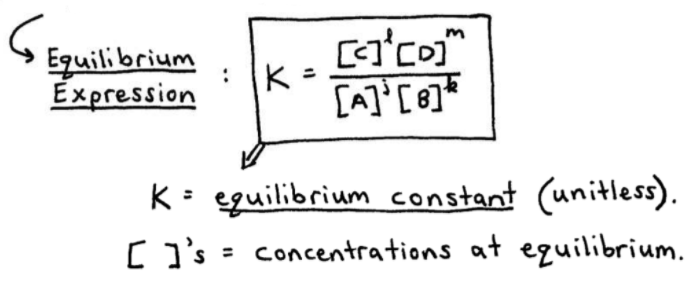
![SOLVED:Calculate K eq for the equilibrium in Practice Problem 1 c on page 601 using the data [CO]=0.0613 mol / L, [H2]=0.1839 mol / L, [CH4]=0.0387 mol / L, and [H2 O]=0.0387 mol / L. SOLVED:Calculate K eq for the equilibrium in Practice Problem 1 c on page 601 using the data [CO]=0.0613 mol / L, [H2]=0.1839 mol / L, [CH4]=0.0387 mol / L, and [H2 O]=0.0387 mol / L.](https://cdn.numerade.com/previews/8e08ded6-a43b-41e1-a20a-1f0a87128134_large.jpg)
SOLVED:Calculate K eq for the equilibrium in Practice Problem 1 c on page 601 using the data [CO]=0.0613 mol / L, [H2]=0.1839 mol / L, [CH4]=0.0387 mol / L, and [H2 O]=0.0387 mol / L.

Calculate the equation constant for the reaction: H2 (g) + CO2 (g) H2O (g) + CO at 1395 K, if the equilibrium constants at 1395 K for the following are 2H2O (g)

How to Calculate Equilibrium Constant K Value Practice Problems & Exampled Explained Step by Step - YouTube
![SOLVED:Calculate K eq for the equilibrium in Practice Problem 1a on page 601 using the data [N2 O4]=0.0185 mol / L and [NO2]=0.0627 mol / L. SOLVED:Calculate K eq for the equilibrium in Practice Problem 1a on page 601 using the data [N2 O4]=0.0185 mol / L and [NO2]=0.0627 mol / L.](https://cdn.numerade.com/previews/86b6e4a5-8685-4656-aeb3-aef2c81a41f9_large.jpg)
SOLVED:Calculate K eq for the equilibrium in Practice Problem 1a on page 601 using the data [N2 O4]=0.0185 mol / L and [NO2]=0.0627 mol / L.

Calculate the equilibrium constant for the the reaction given below at 400K, if Delta H ^(@) = 77.2 kJ mol ^(-1) and Delta S ^(@) = 122JK ^(-1) mol ^(-1) , PCl (s (s)) to PCl (3 (g)) + Cl (2 (g))
![Calculate Kp for the reaction, C(s) + H2O(g) CO(g) + H2(g) at 990 K if the equilibrium concentration are as follows : [H2O] = 1.10 M, [CO] = [H2] = 0.2 M, Calculate Kp for the reaction, C(s) + H2O(g) CO(g) + H2(g) at 990 K if the equilibrium concentration are as follows : [H2O] = 1.10 M, [CO] = [H2] = 0.2 M,](https://dwes9vv9u0550.cloudfront.net/images/6653152/88b9752c-8929-4745-bc8f-8705cb2ad9b9.jpg)


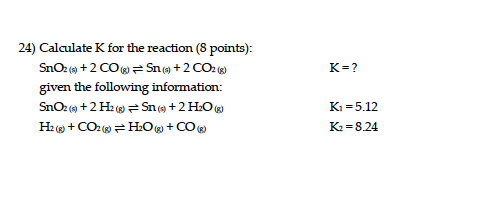
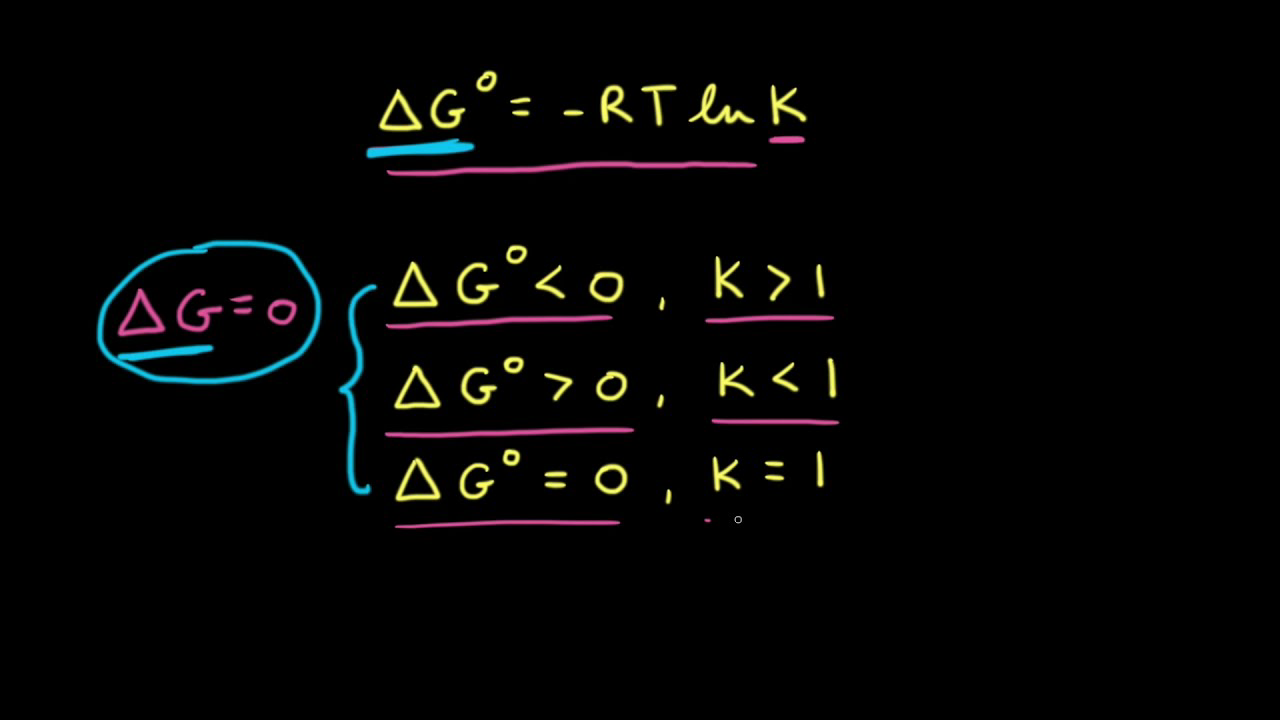
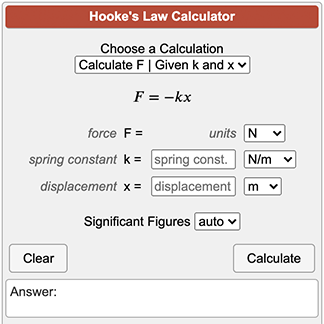

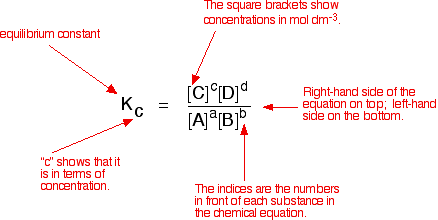

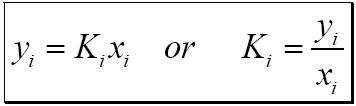

![Calculate K from [EQUILIBRIUM] concentrations 2018 - YouTube Calculate K from [EQUILIBRIUM] concentrations 2018 - YouTube](https://i.ytimg.com/vi/8l1HCaVLgVI/maxresdefault.jpg)